Résumés Année 1994
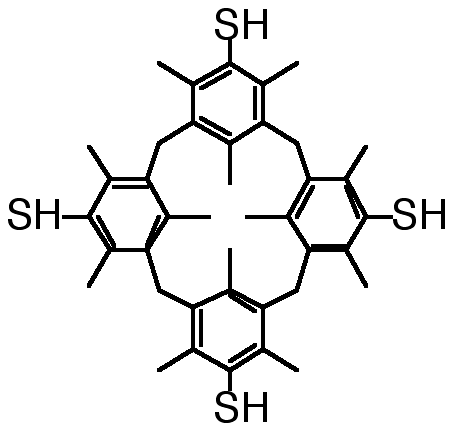
"Synthesis of a 1,3-Alternate Tetramercapto[1.1.1.1]Metacyclophane"
X. DELAIGUE, M. W. HOSSEINI, Tetrahedron Lett. , 8111-8112, 35 1994.
Abstract : 1,3-alternate 3,5,7,10,12,14,17,19,21,24,26,28-dodecamethyl[1.1.1.1]metacyclophane-4,11,18,25-tetrathiol was prepared in high yield in three steps starting from the tetrahydroxy metacyclophane.
 |  |
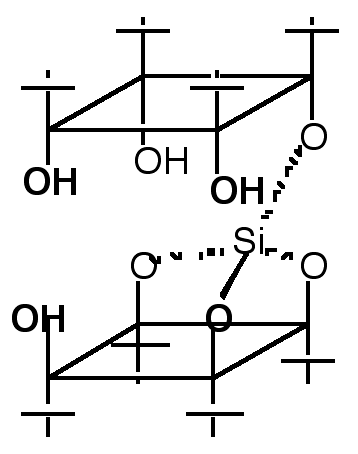
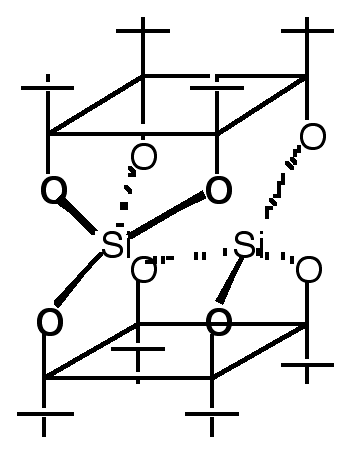
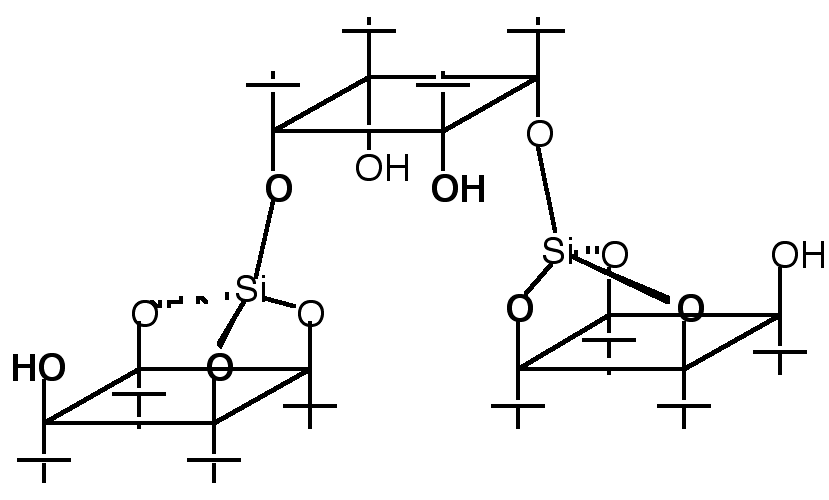
"Multicavitand III: Synthesis and NMR Studies of a Tri-directional Koiland Composed of Three p-tert-butylcalix[4]arene Units Fused by two Silicon Atoms"
O. FELIX, M. W. HOSSEINI, A. DE CIAN, J. FISCHER, Tetrahedron Lett., 1997, 38, 1755-1758.
Abstract : The synthesis of a divergent trireceptor composed of three hydrophobic cavities was achieved by fusing three p-tert-butylcalix[4]arene units in their cone conformation with two silicon atoms. Complete structural analysis was possible using multidimensional NMR techniques. Variable temperature NMR studies between 40°C and -90°C revealed that all three calix units were in their cone conformation and that the only conformational change obser ve d was a rotation around the silicon-oxygen bonds with a DG‡ of ca. 46 kJ mol-1.
 |  |  |
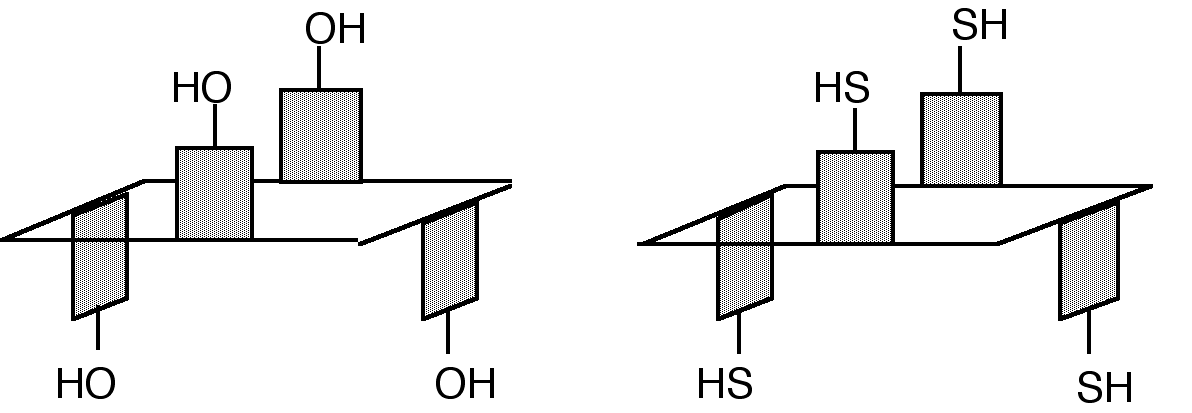
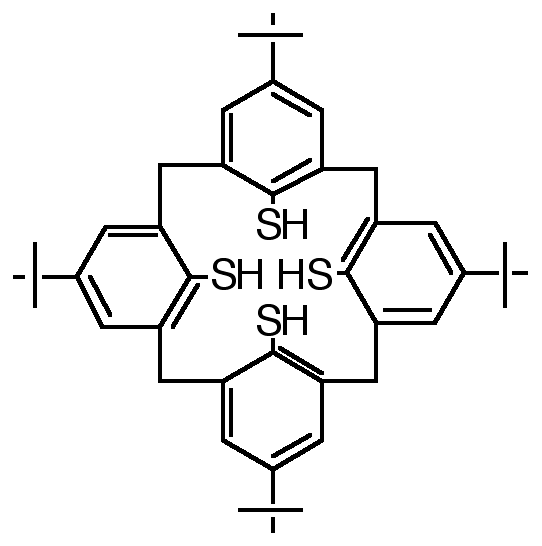
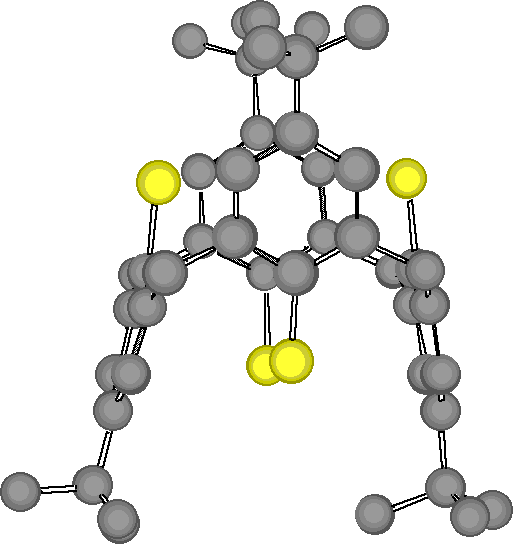
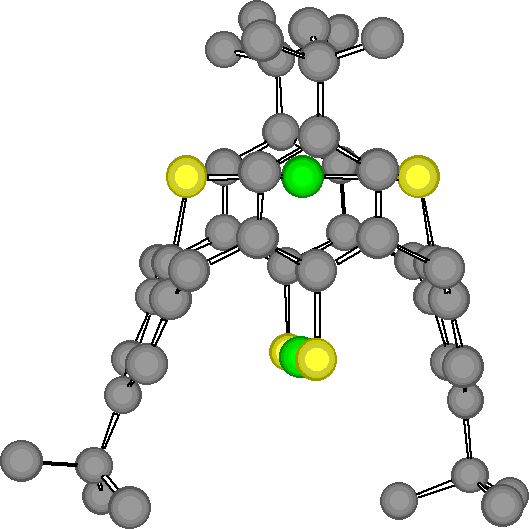
"Exoditopic Receptor I: Synthesis and Structural Studies on p-tert-butyltetramercapto- calix[4]arene and its Mercury Complexes"
X. DELAIGUE, J. McB. HARROWFIELD, M. W. HOSSEINI, A. DE CIAN, J. FISCHER, N. KYRITSAKAS, J. C. S. Chem. Commun. , 1579-1580, 1994.
Abstract : Reaction of mercury (II) acetate with the 1,3-alternate conformer of p-tertbutyltetramercaptocalix[4]arene, obtained by a highly efficient synthesis starting from p-tertbutylcalix[4]arene, leads to a binuclear complex in which both Hg ions are linearly coordinated to two arenethiolate groups.
 |  |  |
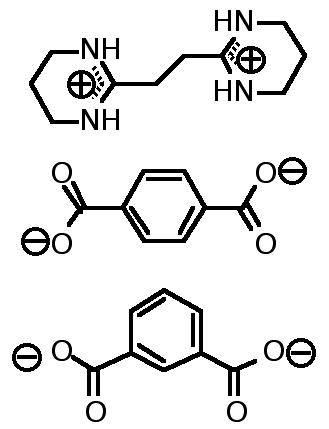
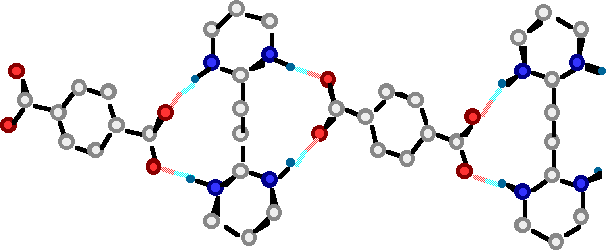
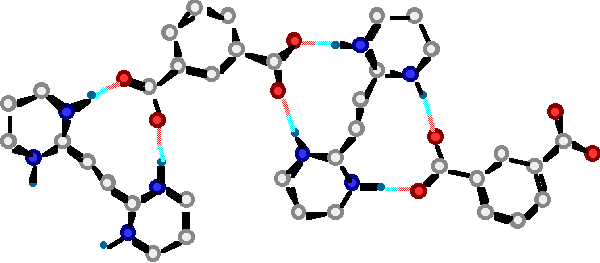
"A molecular Approach to the Solid State Synthesis: Prediction and Synthesis of self-assembled Infinite Rods"
M. W. HOSSEINI, R. RUPPERT, P. SCHAEFFER, A; DE CIAN, N; KYRITSAKAS, J; FISCHER, J. C. S. Chem. Commun. , 2135-2136, 1994.
Abstract :
 |  |  |
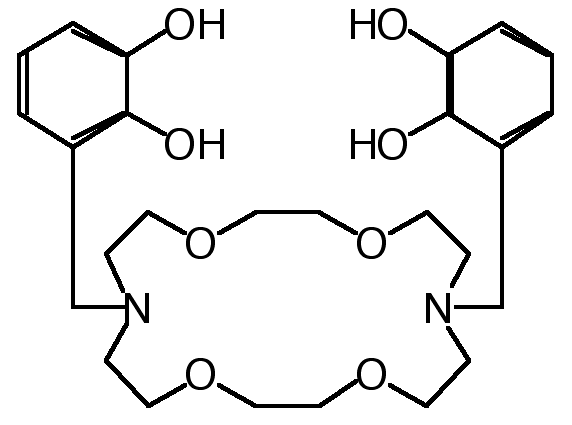
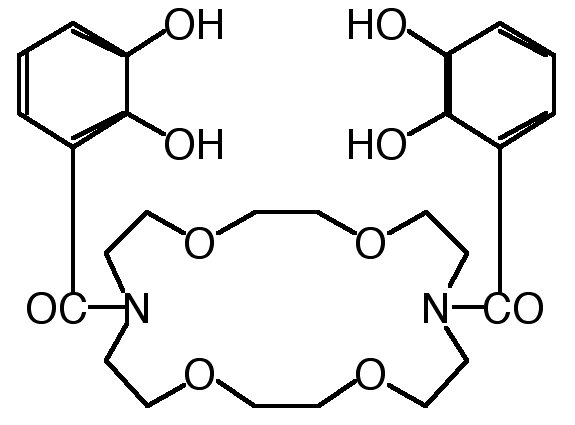
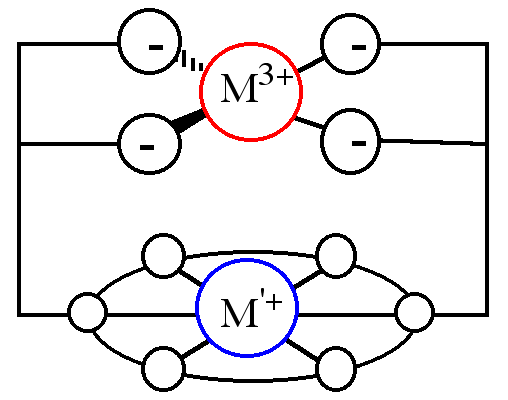
"Synthesis of Macrocyclic Ditopic Receptors Designed for Simultaneous Binding of Alkaline and Transition Metal Cations"
E. GRAF, M. W. HOSSEINI, R. RUPPERT, Tetrahedron Lett. , 7779-7782, 35 , 1994.
Abstract : The synthesis of diazatetraoxacyclooctadecane derivatives bearing two catechol groups was achieved in good yield. The binucleating ligands were designed to bind both soft and hard cations simultaneously.
 |  |  |
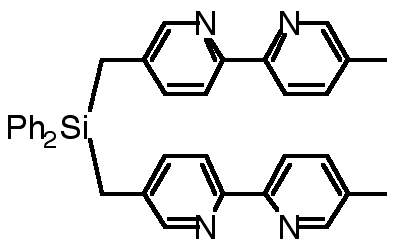
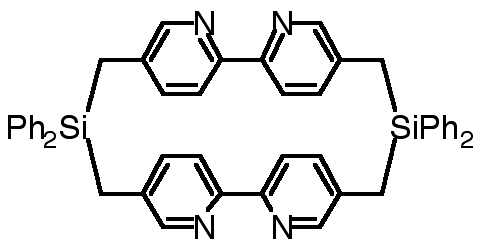
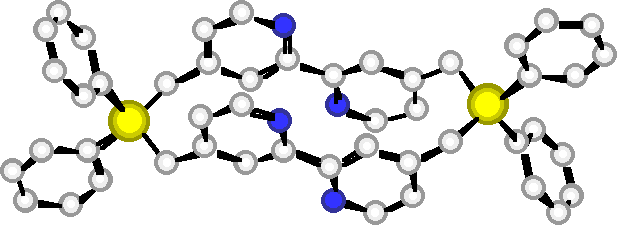
"Exoditopic Receptors II: Synthesis and X-ray Crystal Structure of a Disilamacrocycle Bearing Two Bipyridine Units"
C. KAES, M. W. HOSSEINI, R. RUPPERT, A. DE CIAN, J. FISCHER, Tetrahedron Lett. , 7233-7236, 35 , 1994.
Abstract : The synthesis of a macrocyclic compound composed of two 2,2'-bipyridines interconnected at the 4 and 4' positions by two -CH2SiCH2- fragments was achieved and its structure was established in the solid state by X-ray crystallography.
 |  |  |
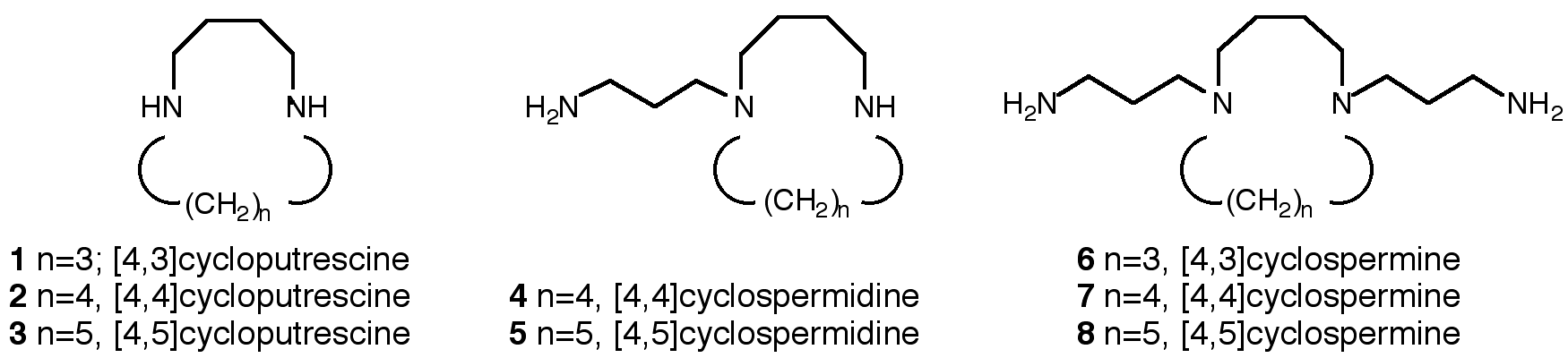
"Cyclopolyamines: Synthesis of Cyclospermidines and Cyclospermines Analogues of Spermidine and Spermine"
G. BRAND, M. W. HOSSEINI, R. RUPPERT, Tetrahedron Lett. , 8609-8612, 35, 1994.
Abstract : The synthesis of a series of cyclic analogues of the naturally occurring polyamines putrescine, spermidine and spermine was achieved. The cyclic analogues of putrescine named cycloputrescine [4, n] were composed of cycloputrescine in which the two primary amines we re connected with a polymethylene chain of variable length (n = 3, 4, 5). Cyclospermidines [4, n] and Cyclospermines [4, n] were comp osed of a cycloputrescine [4,n] moiety mono- or di-functionalized with an aminopropyl chain respectively.